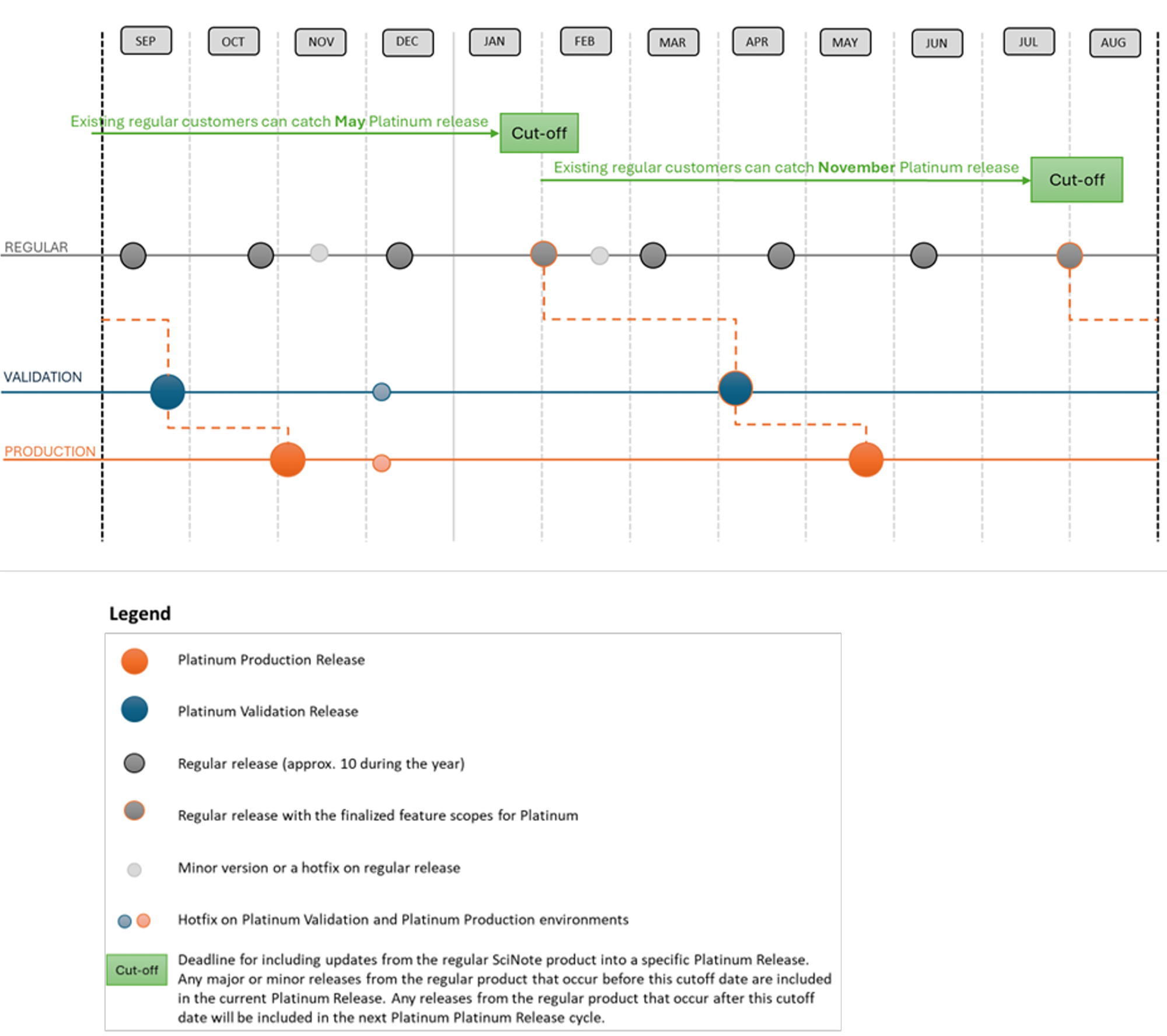
SciNote provides a dedicated validation environment that allows organizations to safely verify and validate system updates before they are deployed to the working environment.
This gives your team a safe space to evaluate new software releases without impacting daily laboratory workflows. This approach ensures that all updates are properly assessed and validated before they reach the production environment.
By separating validation from production, teams can ensure system integrity, regulatory compliance, and uninterrupted laboratory operations.
Using a validation environment ensures that updates can be thoroughly evaluated and verified without affecting ongoing laboratory work or critical data.
Early access allows teams to:
- Review new features and system improvements
- Perform internal validation procedures
- Test workflows and integrations
- Update Standard Operating Procedures (SOPs)
- Ensure compliance with internal quality requirements