Streamlining Safety: The Essential Guide to Safety Data Sheets (SDS) Management
![]() 7 min read
7 min read
In the dynamic landscape of scientific research and development, laboratories continuously strive for innovation while shouldering the critical responsibility of safely handling hazardous substances. Central to ensuring safety and compliance are Safety Data Sheets (SDS), which offer indispensable information on chemical properties, hazards, and necessary precautions. Given their regulatory complexity and critical role, how can labs efficiently manage SDS to enhance compliance and operational effectiveness?
This comprehensive guide explores the fundamentals of SDS management, including regional requirements, best practices, and the benefits of integrating SDS management into systems such as ELNs and LIMS..
Content
The Importance of SDS in Laboratory Safety
Safety Data Sheets serve as a primary communication tool, detailing the risks and safe handling practices of hazardous chemicals. They play a critical role in ensuring laboratory safety and regulatory compliance. An SDS is required for any chemical classified as hazardous under specific regional regulations. Comprising a comprehensive 16-section format, SDSs offer vital information ranging from chemical identification and hazards to disposal considerations. Ensuring that these documents are accurate and readily accessible is essential for safeguarding laboratory personnel and operations.
Global Regulatory Requirements for SDS Compliance
Different global regions impose specific regulations regarding SDS compliance:
European Union:
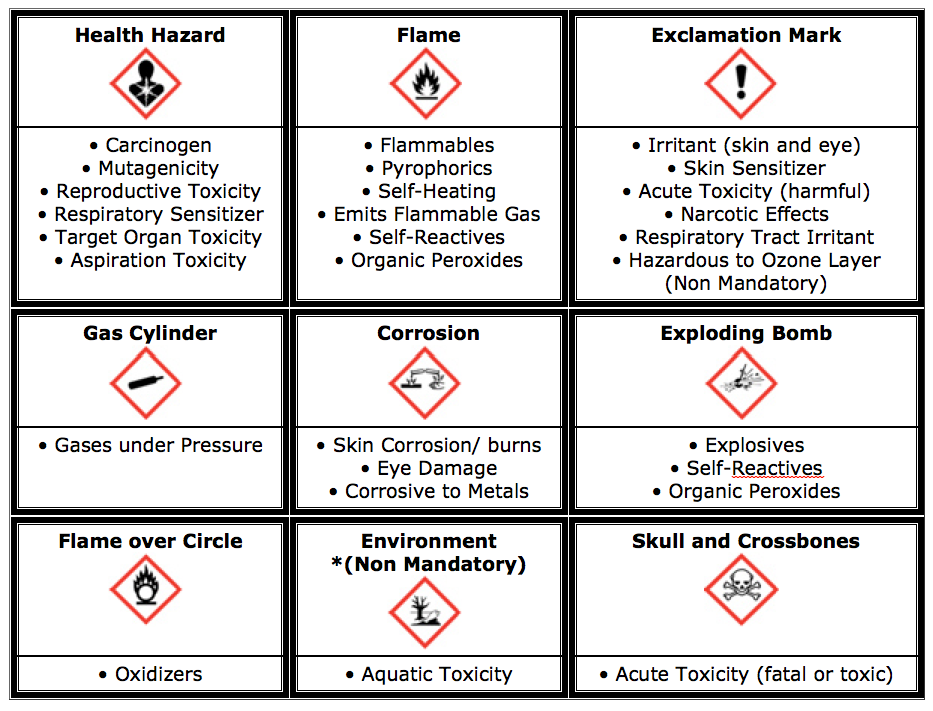
Regulation (EU) 2020/878 mandates that SDSs must adhere to a standardized 16-section format as defined in Annex II of REACH. They must be available in the official language(s) of the member state and be updated quickly with any new hazard information or the addition of a Unique Formula Identifier (UFI). Important to note is the German Hazardous Substances Ordinance (GefStoffV), which supplements these EU regulations specifically in the area of occupational safety. While REACH/CLP regulates the product on the market, GefStoffV focuses on safe handling in the workplace, emphasizing hazard assessments, protective measures, and training. The European Chemicals Agency (ECHA) coordinates the implementation of these regulations, ensuring that SDSs comply with REACH via the GHS system and uniform hazard pictograms (red and white diamonds) that replace older orange symbols.
United States:
The OSHA 2012 Hazard Communication Standard (HCS) requires SDSs to follow a similar structured format and be accessible to employees in English. Updates are mandated within three months when new hazard data emerges.
Canada:
WHMIS 2015 dictates SDSs be available in both English and French, requiring comprehensive sections and updates within 90 days upon acquiring new information.
Challenges in Traditional SDS Management
Traditional SDS management faces several obstacles:
- Fragmented Systems: SDS management often involves disconnected systems for storage and access, leading to inefficiencies and potential gaps in communication.
- Manual Updates: Continuous manual tracking and updating of SDS is labor-intensive and prone to errors, risking non-compliance and outdated safety information.
- Access Issues: Ensuring immediate access to the most current SDSs for all staff, particularly during emergencies, is often inadequately addressed.
Best Practices for Effective SDS Management
Implementing structured practices can transform SDS management:
Centralized Document Management
Utilize a digital platform to centralize SDS storage, attaching documents to logged chemicals in the inventory for efficient retrieval and compliance checks.
Location Management and Access Control
Manage inventory locations from building to the box level, and implement access permissions to ensure only authorized personnel can view sensitive data.
Regular Reviews and Updates
Schedule periodic reviews to incorporate new hazard information and ensure alignment with current regulatory standards.
Accessibility and Language Management
Ensure SDSs are available in required languages and can be accessed by all employees, meeting regional regulatory requirements.
Automated Alerts and Notifications
Deploy systems that provide automated alerts for new updates to keep safety information current.
Comprehensive Labeling with QR Codes or Pictograms
Generate labels with QR codes and pictograms to facilitate quick chemical identification and enhance safety communication. QR codes can link directly to digital SDS versions, ensuring instant access to the most up-to-date information.
The Transformative Role of Digital Solutions in SDS Management
Digital solutions provide robust tools that streamline the management of SDS:
- Document Upload and Management:
Easily upload and manage SDS documents, ensuring they are linked to corresponding inventory items (chemicals) for quick access during audits or emergencies. - Improved Accessibility and Permissions:
Systems can enhance accessibility by allowing employees immediate access to the latest SDS updates.
Benefits of Integrating SDS with ELN and LIMS
Incorporating SDS management within Electronic Lab Notebooks (ELN) or Laboratory Information Management Systems (LIMS) such as SciNote delivers multiple advantages:
- Enhanced Data Management:
Integrates SDS management with inventory tracking and experimental data, providing comprehensive oversight and streamlined operations. - Improved Compliance and Safety:
Adheres to regulatory standards rigorously, minimizing the risk of non-compliance and enhancing laboratory safety. - Streamlined Operations:
Reduces administrative burdens, allowing labs to focus on core research activities by integrating SDS management seamlessly with daily operations.
Conclusion
Safety Data Sheets are indispensable for ensuring chemical safety and regulatory compliance in laboratories. By adopting a structured approach and integrating SDS management into digital systems like ELN and LIMS, labs can significantly enhance efficiency, compliance, and safety. Embrace the transition towards streamlined safety management to experience the comprehensive benefits of integrating these vital systems into your lab operations.
For a deeper dive into innovative SDS management solutions, download the SciNote Inventory Management PDF or book a demo today to see how this integration can optimize your lab’s safety and efficiency.